SUPRATENTORIAL BRAIN TUMORS
John R. Hesselink, MD, FACR and Richard J. Hicks, MD
In the diagnostic work-up of intracranial tumors, the primary goals of the imaging studies are to detect the abnormality, localize and determine its extent, characterize the lesion, and provide a list of differential diagnoses or, if possible, the specific diagnosis. Correlative studies have proved that MR is more sensitive than CT for detecting intracranial masses. Moreover, the multiplanar capability of MR is very helpful to determine the anatomic site of origin of lesions and to demarcate extension into adjacent compartments and brain structures. The superior contrast resolution of MR displays the different components of lesions more clearly. MR can assess the vascularity of lesions without contrast infusion. On the other hand, CT detects calcification far better than MR, a useful finding for differential diagnosis. Gradient-echo techniques improve MR detection of calcification by accentuating the diamagnetic susceptibility properties of calcium salts, but the observed low signal on T2-weighted images is nonspecific, in that any accompanying paramagnetic ions would produce the same effect.

Contrast enhancement with gadolinium
increases both the sensitivity and specificity of
MR.
![]() Gadolinium is a blood-brain barrier (BBB) contrast agent like iodinated agents for CT. It does
not cross the intact BBB, but when the BBB is absent or deficient, gadolinium enters the interstitial
space to produce enhancement (increased signal) on T1-weighted images. All the collective
knowledge learned from contrast-enhanced CT can be applied directly to the gadolinium-enhanced
MR images. Although the enhancement patterns are not tumor specific, the additional information
is often helpful for diagnosis. Lesions can be classified as homogeneous or heterogeneous, and
necrotic and cystic components are seen more clearly. The margins of enhancement provide a gross
measure of tumor extension.
Gadolinium is a blood-brain barrier (BBB) contrast agent like iodinated agents for CT. It does
not cross the intact BBB, but when the BBB is absent or deficient, gadolinium enters the interstitial
space to produce enhancement (increased signal) on T1-weighted images. All the collective
knowledge learned from contrast-enhanced CT can be applied directly to the gadolinium-enhanced
MR images. Although the enhancement patterns are not tumor specific, the additional information
is often helpful for diagnosis. Lesions can be classified as homogeneous or heterogeneous, and
necrotic and cystic components are seen more clearly. The margins of enhancement provide a gross
measure of tumor extension.
![]() Contrast MR is particularly
valuable for extra-axial tumors because they tend to be
isointense to the brain on plain scan.
Contrast MR is particularly
valuable for extra-axial tumors because they tend to be
isointense to the brain on plain scan.
CEREBRAL GLIOMAS
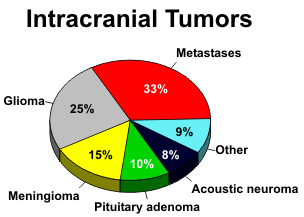
Gliomas are malignant tumors of the glial cells of the brain and account for 30-40% of all primary intracranial tumors. They occur predominantly in the cerebral hemispheres, but the brain stem and cerebellum are frequent locations in children, and they are also found in the spinal cord. The peak incidence is during middle adult life, when patients present with seizures or symptoms related to the location of the gliomas and the brain structures involved.
Astrocytomas are graded according to their histologic appearance. Grade 1 astrocytomas have well-differentiated astrocytes and well-defined margins. The clinical course often proceeds over many years and complete cures are possible. The pilocytic variant is a low-grade tumor with a distinct capsule that is commonly found in children. The giant cell astrocytoma is a specialized tumor that develops from pre-existing hamartomas in patients with tuberous sclerosis. Grade 2 astrocytomas are well-differentiated but diffusely infiltrating tumors. The fibrillary type is most common, and although initially benign, they may evolve into a higher grade tumor over time. This changing character of gliomas makes histological classification difficult from sample biopsies, because different parts of the tumor often exhibit varying degrees of malignancy. The higher grade astrocytomas are very cellular and pleomorphic. Anaplastic astrocytomas (Grade 3) are very aggressive tumors, readily infiltrate adjacent brain structures, and have a uniformly poor prognosis. Glioblastoma multiforme (Grade 4) has the added histologic features of endothelial proliferation and necrosis. Multicentric foci of tumor may be seen in 4 to 6% of glioblastomas. Gliomatosis cerebri is an unusual condition with diffuse contiguous involvement of multiple lobes of the brain.
Oligodendrogliomas are the most benign of the gliomas. Calcification is common, and they
occur predominantly in the frontal lobes.
![]() The mixed neuronal and glial tumors are found mostly
in children and young adults. They are slow-growing and are found predominantly in the temporal
lobes and around the third ventricle. Intratumoral cysts and calcification are common.
The mixed neuronal and glial tumors are found mostly
in children and young adults. They are slow-growing and are found predominantly in the temporal
lobes and around the third ventricle. Intratumoral cysts and calcification are common.
![]()
The common signal characteristics of intra-axial tumors include high signal intensity on T2-weighted images and low signal on T1-weighted images, unless fat or hemorrhage is present. Fat and subacute hemorrhage (methemoglobin) exhibit high signal on T1-weighted images, and acute hemorrhage (deoxyhemoglobin) and chronic hemorrhage (hemosiderin/ferritin) show low signal intensity on T2-weighted scans. Gliomas have poorly defined margins on plain MR. They infiltrate along white matter fiber tracts, and the deeper lesions have a propensity to extend across the corpus callosum into the opposite hemisphere. They are often quite large by the time of clinical presentation.
The higher grade gliomas, particularly glioblastomas, appear heterogeneous due to central
necrosis with cellular debris, fluid, and hemorrhage. Peritumoral edema and mass effect are common
features. Following injection of gadolinium, T1-weighted images show irregular ring enhancement,
with nodularity and nonenhancing necrotic foci.
![]() As mentioned above, gliomas are infiltrative lesions,
and microscopic fingers of tumor usually extend beyond the margin of enhancement. Enhanced scans
are particularly helpful to outline subependymal spread of tumor along a ventricular surface, as well
as leptomeningeal involvement.
As mentioned above, gliomas are infiltrative lesions,
and microscopic fingers of tumor usually extend beyond the margin of enhancement. Enhanced scans
are particularly helpful to outline subependymal spread of tumor along a ventricular surface, as well
as leptomeningeal involvement.
![]() Although highly malignant, anaplastic astrocytomas may or may not
exhibit breakdown of the blood-brain barrier. In general, the presence or lack of enhancement alone
is not helpful in grading astrocytomas.
Although highly malignant, anaplastic astrocytomas may or may not
exhibit breakdown of the blood-brain barrier. In general, the presence or lack of enhancement alone
is not helpful in grading astrocytomas.
The lower grade astrocytomas tend to be more homogeneous without central necrosis. Large cystic components may be present. The cysts have smooth walls, and the fluid is of uniform signal, to distinguish them from necrosis. Enhancement is variable, depending on the integrity of the blood-brain barrier.
Perfusion imaging has shown promise as a technique for determining the grade of intracranial
mass lesions. Perfusion imaging relies on a first-pass susceptibility-related signal loss on
T2*-weighted images, from which relative cerebral blood flow and volume can be calculated. Several
studies have shown a correlation between relative cerebral blood volume and tumor grade, likely due
to the relationship of blood volume to vascular proliferation in high-grade gliomas.
![]()
MR Spectroscopy
MR spectroscopy provides a measure of brain chemistry and can help characterize tumors and and grade the degree of malignancy. As a general rule, as malignancy increases, NAA and creatine decrease, and choline, lactate, and lipids increase. NAA decreases as tumor growth displaces or destroys neurons. Very malignant tumors have high metabolic activity and deplete the energy stores, resulting in reduced creatine. Very hypercellular tumors with rapid growth elevate choline. Lipids are found in necrotic portions of tumors, and lactate appears when tumors outgrow their blood supply and start utilizing anaerobic glycolysis. To get an accurate assessment of the tumor chemistry, the spectroscopic voxel should be placed over an enhancing region of the tumor, avoiding areas of necrosis, hemorrhage, calcification, or cysts.
Multi-voxel spectroscopy is best to detect infiltration of malignant cells beyond the enhancing
margins of tumors. Particularly in the case of cerebral glioma, elevated choline levels are frequently
detected in edematous regions of the brain outside the enhancing mass.
![]() Finally, MRS can direct the
surgeon to the most metabolically active part of the tumor for biopsy to obtain accurate grading of
the malignancy.
Finally, MRS can direct the
surgeon to the most metabolically active part of the tumor for biopsy to obtain accurate grading of
the malignancy.

A common clinical problem is distinguishing tumor recurrence from radiation effects several months following surgery and radiation therapy. Elevated choline is a marker for recurrent tumor. Radiation change generally exhibits low NAA, creatine, and choline on spectroscopy. If radiation necrosis is present, the spectrum may reveal elevated lipids and lactate.
MRS cannot always distinguish primary and secondary
tumors of the brain from one another. As mentioned above, one
key feature of gliomas is elevated choline beyond the margin of
enhancement due to infiltration of tumor into the adjacent brain
tissue. Most non-glial tumors have little or no NAA. Elevated
alanine at 1.48 ppm is a signature of meningiomas. They also
have no NAA, very low creatine, and elevated glutamates.
![]()
LYMPHOMA
Primary malignant lymphoma is a non-Hodgkin's lymphoma that occurs in the brain in the absence of systemic involvement. These tumors are highly cellular and grow rapidly. Favorite sites include the deeper parts of the frontal and parietal lobes, basal ganglia, and hypothalamus. Most occur in patients who are immunocompromised secondary to chemotherapy or acquired immunodeficiency syndrome (AIDS) or in organ transplant recipients who are on immunosuppressant drugs. Cerebral lymphomas are very radiosensitive and respond dramatically to steroid therapy.
Lymphomas typically appear as homogeneous, slightly high signal to isointense masses deep
within the brain on T2-weighted images. The observed mild T2 prolongation is probably related to
dense cell packing within these tumors, leaving relatively little interstitial space for accumulation of
water. They are frequently found in close proximity to the corpus callosum and have a propensity
to extend across the corpus callosum into the opposite hemisphere, a feature that mimics
glioblastoma. Multiple lesions are present in as many as 50%. Despite their rapid growth, central
necrosis is uncommon. They are associated with only a mild or moderate amount of peritumoral
edema. By time of presentation they can be quite large and yet produce relatively little mass effect,
a feature that sets lymphoma apart from glioblastoma and metastases. Intratumoral cysts and
hemorrhage are unusual. Most lymphomas show bright homogeneous contrast enhancement.
![]()
The pattern is modified somewhat in AIDS patients. Multiplicity seems to be more common.
Moreover, lymphomas exhibit more aggressive behavior and readily outgrow their blood supply. As
a result, central necrosis and ring enhancement are often seen in lymphomatous masses in AIDS
patients.
![]() On MR spectroscopy, lymphomas exhibit elevated choline little or no NAA.
On MR spectroscopy, lymphomas exhibit elevated choline little or no NAA.
METASTATIC DISEASE
Metastases to the brain occur by hematogenous spread, and multiple lesions are found in 70% of cases. The most common primaries are lung, breast, and melanoma, in that order of frequency. Other potential sources include the gastrointestinal tract, kidney, and thyroid. Metastases from other locations are uncommon. Clinical symptoms are nonspecific and no different from primary brain tumors. If a parenchymal lesion breaks through the cortex, tumor can extend and seed along the leptomeninges.
Metastatic lesions can be found anywhere in the brain but a favorite site is near the brain surface at the corticomedullary junction of both the cerebrum and cerebellum. They are hyperintense on plain T2-weighted images. Areas of necrosis are prevalent in the larger lesions, accounting for their heterogeneous internal texture. Peritumoral edema is a prominent feature, but multiplicity is the most helpful sign to suggest metastatic disease as the likely diagnosis. Correlative studies have shown MR to be more sensitive than CT for detecting metastases, particularly lesions near the base of the brain and in the posterior fossa. One limitation of plain MR is the frequency of periventricular white matter hyperintensities found in the same older age group at risk for metastatic disease.
Gadolinium enhanced MR has resulted in improved delineation of metastatic disease
compared with nonenhanced scans. Moderate to marked enhancement is the rule, nodular for the
smaller lesions and ringlike with central nonenhancing areas for the larger ones.
![]() Controlled clinical
trials have also shown that contrast-enhanced MR is more sensitive than both plain MR and contrast-enhanced CT for detecting cerebral metastases. In patients with a known primary, T1-weighted
enhanced MR is probably sufficient to screen the brain for metastatic disease.
Controlled clinical
trials have also shown that contrast-enhanced MR is more sensitive than both plain MR and contrast-enhanced CT for detecting cerebral metastases. In patients with a known primary, T1-weighted
enhanced MR is probably sufficient to screen the brain for metastatic disease.
Hemorrhage is present in 3 to 14% of brain metastases, mainly in melanoma, choriocarcinoma, renal cell carcinoma, bronchogenic carcinoma, and thyroid carcinoma. The presence of nonhemorrhagic tissue and pronounced surrounding vasogenic edema are clues to the underlying neoplasm.
Metastatic melanoma has been a topic of special interest in the MR literature because of the
presence of paramagnetic, stable free radicals within melanin. The MR appearance is variable
depending on the histology of the melanoma and the components of hemoglobin. Most are
hyperintense to white matter on T1-weighted scans and hypointense on T2-weighted scans. Atlas
and coworkers
![]() observed three distinct signal intensity patterns. Nonhemorrhagic melanotic
melanoma was markedly hyperintense on T1-weighted images and isointense or mildly hypointense
on T2-weighted images. Nonhemorrhagic amelanotic melanoma appeared isointense or slightly
hypointense on T1-weighted scans and isointense or slightly hyperintense on T2-weighted scans. The
signal pattern for hemorrhagic melanoma was variable depending on the components of hemoglobin.
Some uncertainty remains as to whether the predominant effect on signal intensity within melanomas
is due to stable free radicals, chelated metal ions, or hemoglobin.
observed three distinct signal intensity patterns. Nonhemorrhagic melanotic
melanoma was markedly hyperintense on T1-weighted images and isointense or mildly hypointense
on T2-weighted images. Nonhemorrhagic amelanotic melanoma appeared isointense or slightly
hypointense on T1-weighted scans and isointense or slightly hyperintense on T2-weighted scans. The
signal pattern for hemorrhagic melanoma was variable depending on the components of hemoglobin.
Some uncertainty remains as to whether the predominant effect on signal intensity within melanomas
is due to stable free radicals, chelated metal ions, or hemoglobin.

INTRAVENTRICULAR TUMORS
The intraventricular location is unique in that many of the tumor types are more commonly associated with extra-axial locations. Patients often present with obstructive hydrocephalus. Most intraventricular tumors are relatively benign and have well-defined margins. As they grow, the tumors expand the ventricle of origin. With malignant degeneration, extension into the brain parenchymal occurs. The primary blood supply to intraventricular lesions is derived from the choroidal arteries.
MENINGIOMA
Meningiomas account for 15% of all intracranial tumors and are the most common extra-axial tumor. They originate from the dura or arachnoid and occur in middle-aged adults. Women are affected twice as often as men. Meningiomas are well-differentiated, benign, and encapsulated lesions that indent the brain as they enlarge. They grow slowly and may be present for many years before producing symptoms. The histologic picture shows cells of uniform size that tend to form whorls or psammoma bodies.
The parasagittal region is the most frequent site for meningiomas, followed by the sphenoid wings, parasellar region, olfactory groove, cerebello-pontine angle, and rarely the intraventricular region. Meningiomas often induce an osteoblastic reaction in the adjacent bone, resulting in a characteristic focal hyperostosis. They are also hypervascular, receiving their blood supply predominantly from dural vessels.
Most meningiomas are isointense with cortex on T1- and T2-weighted images. A heterogeneous internal texture is found in all but the smallest meningiomas. The mottled pattern is likely due to a combination of flow void from vascularity, focal calcification, small cystic foci, and entrapped CSF spaces. Hemorrhage is not a common feature. An interface between the brain and lesion is often present, representing a CSF cleft, a vascular rim, or a dural margin. MR has special advantages over CT in assessing venous sinus involvement and arterial encasement. Occasionally, a densely calcified meningioma is encountered that is distinctly hypointense on all pulse sequences.
Meningiomas show intense enhancement with gadolinium and are sharply circumscribed.
![]() They have a characteristic broad base of attachment against a dural surface. Associated hyperostosis
may result in thickening of low signal bone as well as diminished signal from the diploic spaces.
Although meningiomas are not invasive, vasogenic edema is present in the adjacent brain in 30% of
cases. Contrast scans are especially helpful for imaging the en plaque meningiomas that occur at the
skull base. MR spectroscopy shows elevated alanine and glutamates, no NAA, and markedly
decreased creatine.
They have a characteristic broad base of attachment against a dural surface. Associated hyperostosis
may result in thickening of low signal bone as well as diminished signal from the diploic spaces.
Although meningiomas are not invasive, vasogenic edema is present in the adjacent brain in 30% of
cases. Contrast scans are especially helpful for imaging the en plaque meningiomas that occur at the
skull base. MR spectroscopy shows elevated alanine and glutamates, no NAA, and markedly
decreased creatine.
PINEAL REGION TUMORS
Tumors in the pineal region can be classified into three major groups based on their origin:
germ cell, pineal parenchyma, and parapineal. Germinoma is the least differentiated of the germ cell
group. It occurs in children and young adults and accounts for more than 50% of all pineal region
tumors. The other germ cell tumors include embryonal carcinoma, yolk-sac tumor, and
choriocarcinoma. Differentiation along three germ layers results in a teratoma. The true
pinealomas consist of pineoblastoma and pineocytoma. Pineoblastoma is an embryonal tumor of
neuroectoderm, related to neuroblastoma and medulloblastoma, and is found primarily in young
children. Pineocytomas are less cellular and exhibit benign behavior. The parapineal lesions include
gliomas of the tectum and posterior third ventricle, meningiomas arising within the quadrigeminal
cistern, and developmental cysts (epidermoid, dermoid, arachnoid cyst).
![]()
The clinical expression of these tumors is usually related to mass effect upon adjacent brain structures. Hydrocephalus secondary to aqueductal obstruction is a common presentation. Compression of the tectum of the midbrain can produce paralysis of upward gaze, the classic Parinaud's syndrome. Germinomas and gliomas have a propensity to grow into the third ventricle and compress the hypothalamus, resulting in endocrine dysfunction. Dissemination through the CSF pathways is a known complication of pineoblastoma and germinoma.
Pineal germinomas and primary pineal tumors are most often isointense with the brain on T1-
and T2-weighted images. A few lesions exhibit long T1 and T2, which may correlate with embryonal
cell elements. Despite this relative lack of contrast, with multiplanar imaging plain MR delineates
pineal region masses better than CT, showing the relationships of the tumor to the posterior third
ventricle, vein of Galen, and aqueduct.
![]() These tumors are well defined and enhance to a moderate
degree, usually without central necrosis, cystic change, or hemorrhage. Enhanced scans are essential
to assess CSF spread of tumor. In young patients with germinoma, the difficulty of visualizing
calcium is a disadvantage of MR, as this may be the only evidence of tumor.
These tumors are well defined and enhance to a moderate
degree, usually without central necrosis, cystic change, or hemorrhage. Enhanced scans are essential
to assess CSF spread of tumor. In young patients with germinoma, the difficulty of visualizing
calcium is a disadvantage of MR, as this may be the only evidence of tumor.
![]()
Meningiomas can appear very similar on plain scan, but their intense enhancement may set them apart from other lesions. Gliomas infiltrate the tectum and posterior walls of the third ventricle. They tend to be poorly circumscribed and produce symptoms earlier. Edema is not a consistent finding, and enhancement is variable. Larger gliomas in the splenium of the corpus callosum may present as pineal region masses.
Teratomas are of mixed signal intensity, frequently with calcification. They may also have cystic components and fat. Arachnoid cysts, epidermoid and dermoid tumors can usually be distinguished from other pineal region tumors by their increased signal on T2-weighted images.
Pineal cysts were visualized in 4.3% of normal patients in one MR study.
![]() These apparently
benign lesions are seen best as areas of high signal on intermediate T2-weighted images. They are
not associated with hydrocephalus or a pineal mass and are not clinically significant.
These apparently
benign lesions are seen best as areas of high signal on intermediate T2-weighted images. They are
not associated with hydrocephalus or a pineal mass and are not clinically significant.
BENIGN CYSTIC MASSES
Cystic lesions occur most often in the basal cisterns, a midline location or within the ventricular system. They include arachnoid cyst, dermoid, epidermoid, and neuroepithelial cysts, including colloid cyst. These lesions are interesting in that their MR appearance is quite distinct from solid masses. Their signal characteristics depend to a large extent on the cyst contents, but associated solid components may also have specific features.
Arachnoid Cyst
Arachnoid cysts are CSF-containing cysts that are found in the middle fossa, posterior fossa, suprasellar cistern, or near the vertex. They are benign but slowly grow as they accumulate fluid, compressing normal brain structures. Remodeling of the adjacent skull is an important clue for a benign expansile process.
Arachnoid cysts are smoothly marginated and homogeneous. They are not calcified and do
not enhance. The multiplanar capability of MR is particularly helpful in establishing the exact
location, and the diagnosis is supported by the cyst fluid being isointense with CSF on all pulse
sequences.
![]() The cysts may appear higher signal than CSF on intermediate T2-weighted images. The
exact reason for this is uncertain, although it may reflect dampening of the CSF pulsations that
normally results in signal loss in the ventricles and cisterns. This effect will be less apparent with
pulse sequences that incorporate flow compensation techniques.
The cysts may appear higher signal than CSF on intermediate T2-weighted images. The
exact reason for this is uncertain, although it may reflect dampening of the CSF pulsations that
normally results in signal loss in the ventricles and cisterns. This effect will be less apparent with
pulse sequences that incorporate flow compensation techniques.
Epidermoid Cyst
Epidermoid cysts are referred to as "pearly tumors" because of their glistening white
appearance at surgery. They arise from epithelial cell rests in the basal cisterns. They are benign and
grow slowly along the subarachnoid spaces and into the various crevices found at the base of the
brain. Intradural epidermoids are usually quite large with lobulated outer margins and an
insinuating pattern of growth. They have a heterogeneous texture and variable signal intensity on
MR.
![]() Most are slightly higher signal than CSF on both T1 and T2-weighted images. An occasional
epidermoid has a very short T1 and appears bright on T1-weighted images. The heterogeneous signal
pattern is likely related to varying concentrations of keratin, cholesterol, and water within the cyst,
as well as the proportion of cholesterol and keratin in crystalline form. Calcification is sometimes
present. Epidermoid tumors do not enhance with contrast.
Most are slightly higher signal than CSF on both T1 and T2-weighted images. An occasional
epidermoid has a very short T1 and appears bright on T1-weighted images. The heterogeneous signal
pattern is likely related to varying concentrations of keratin, cholesterol, and water within the cyst,
as well as the proportion of cholesterol and keratin in crystalline form. Calcification is sometimes
present. Epidermoid tumors do not enhance with contrast.
Dermoid Cyst
Dermoid cysts have both dermal and epidermal derivatives, accounting for their more varied histologic and MR appearance. They are primarily midline lesions, occurring in the pineal and suprasellar regions. Dermoids have some distinctive features on MR. They tend to be heterogeneous owing to the multiple cell types within them. Fatty components are common, producing high signal on T1-weighted images. On axial and sagittal scans, a fat-fluid level may be seen, or a level between fat and matted hair within the cyst. Rupture of a dermoid and leakage of cyst contents into a ventricle or subarachnoid space may produce an ependymitis or meningitis, respectively.
Lipomas are also midline lesions and are often associated with partial or complete agenesis of the corpus callosum. Occasionally, an incidental lipoma will be found in the region of the quadrigeminal plate or cerebellopontine angle.
Colloid Cyst
Colloid cysts originate from primitive neuroepithelium within the roof of the anterior third ventricle. They are positioned just posterior to the foramina of Monro between the columns of the fornix. Histologically, they consist of a thin, fibrous capsule with an epithelial lining. The cysts contain a mucinous fluid with variable amounts of proteinaceous debris, blood components, and desquamated cells.
Colloid cysts are smoothly marginated spherical lesions without surrounding brain reaction.
Two signal patterns have been reported on MR scans and correlated with their CT features. Those
that are low density on CT are isointense on T1-weighted images and hyperintense on T2-weighted
images, probably indicating a fluid composition similar to CSF. Most colloid cysts are isodense or
slightly hyperdense on CT. The MR counterpart is a high signal capsule and a hypointense center on
T2-weighted images. The signal characteristics of the fluid depend on the protein content of the cyst
fluid and is similar to that observed in sinonasal secretions.
![]()
Dilatation of the lateral ventricles is a common finding, and the enlargement may be unequal owing to asymmetric positioning of the cyst at the foramina of Monro. The expanding cyst also enlarges the anterior third ventricle, but the posterior third, aqueduct, and fourth ventricle should be normal. Following contrast infusion, colloid cysts may show ring enhancement, due to either enhancement of the cyst wall or choroid plexus draped around the cyst.
REFERENCES