HEAD TRAUMA
John R. Hesselink, MD, FACR
Patients with head trauma constitute a large percentage of the cases referred for neuroimaging. Initially, the role of MR in these patients was considered limited due to the time required for the examination, difficulty in using life-support and monitoring equipment within the scanning room and problems in imaging acute hemorrhage. While some of these problems still remain, MR has come to be used more frequently in these patients, particularly in the subacute period.
The most common head injuries result from blunt or non- penetrating trauma. These
frequently induce a temporary or longer loss of consciousness and the brain may suffer gross
damage despite the lack of a skull fracture or penetrating injury. Skull fractures may serve
to indicate the presence of significant trauma but the absence or presence of a skull fracture
cannot be used to predict the presence or severity of intracranial injury.
![]()
EXTRACEREBRAL HEMORRHAGE
Acute epidural hematomas are often associated with skull fractures and lacerations of the dural vessels, most often meningeal arteries and veins but occasionally a dural sinus. Two-thirds of epidural hematomas are in the temporo-parietal region and they usually have a biconvex or lentiform configuration. Epidurals are limited by the firmer attachment of the dura at the suture margins, but they may cross the midline, especially with superior sagittal sinus lacerations, and they also can bridge the supra- and infratentorial compartments with tears along the torcula and transverse sinuses.
Subdural hematomas, both acute and chronic, are most often caused by bleeding from torn bridging dural veins. Subdural hematomas are less frequently associated with skull fractures, but more frequently associated with parenchymal brain damage. The subdural space is a more freely communicating space and the hematomas form a crescentic shaped layer over the brain surface. Subdural hematomas readily cross suture lines but do not cross the midline. Instead, they extend along the dura of the falx into the interhemispheric fissure and onto the tentorium, which epidurals cannot do. Both epidural and subdural hemorrhages occur within the confined space of the bony calvarium and compress the adjacent brain, often requiring emergency evacuation.
Chronic subdural hematomas are usually related to a slower venous bleed without accompanying cerebral parenchymal injury. A thick,vascular dural membrane forms that can be a source for repeated episodes of hemorrhage. These collections are more often biconvex, rather than the crescentic shape of acute subdural hematomas. The injury leading to a chronic subdural can be relatively minor and may have occurred weeks before presentation. Patients often present with disturbances of mentation and consciousness rather than focal or lateralizing signs. An iatrogenic cause is overshunting or too rapid decompression of chronic hydrocephalus.
Subdural hygromas or effusions consist of collections of CSF in the subdural spaces, presumably due to a traumatic arachnoid tear or they may also develop following ventricular shunting. They may accumulate slowly during the first few days following head trauma, especially in the pediatric population.

Multiple studies have demonstrated improved visualization of extra-axial hemorrhage with
MR compared to CT, largely related to the high conspicuity of hyperintense subacute
hemorrhage (methemoglobin) on T1-weighted images and the multiplanar capabilities of MR.
Coronal images are very helpful for identifying subtemporal collections and hemorrhage
adjacent to the tentorium cerebelli. Chronic subdural hematomas are often isointense with
gray matter on T1-weighted images, probably due to dilution and partial resorption or
breakdown of free methemoglobin. High T1 signal within what otherwise appears to be a
chronic subdural hematoma suggests
rebleeding. Hemosiderin is rarely seen in
subdural hematomas without repeated
episodes of bleeding, due to either low
macrophage activity or removal of
hemosiderin that has formed. The presence of
membranous strands coursing through an
extra-axial collection is additional evidence
for a chronic subdural hematoma. The thick
subdural membranes will also enhance
following contrast infusion.
![]()
BRAIN CONTUSIONS
Damage to the brain parenchyma is a common component of head trauma. The type, location and degree of brain injury is determined to a large extent by the physical properties of the skull and brain. The skull is very hard and rigid, and protects the brain from direct injury. However, the inner table of the skull has roughened edges and ridges of bone along the floor of the anterior cranial fossa, sphenoid wings and petrous ridges that can contuse the brain surface during the compressive forces of trauma.
Injury of the brain parenchyma sets in motion a series of events. Tissue disruption and cell injury are associated with release of vasoactive substances and other byproducts. Subsequent increase in vascular permeability to serum proteins results in a progressive increase in interstitial fluid. Over a period of several days, the edema fluid spreads within the white matter, producing mass effect on adjacent structures and possible further damage. More serious injuries may be associated with vascular disruption and hemorrhage into contusions. Cortical contusion are usually multiple, measuring approximately 2-4 cm in size, and 30% to 50% of lesions are hemorrhagic. Approximately 50% to 75% of cortical contusions involve the frontal and temporal lobes, particularly the lateral surfaces of both lobes and the inferior surface of the frontal lobes.
Varying signal intensity patterns are seen on MR depending on the age and amount of
hemorrhage present. In several studies MR has had a decided advantage over CT in the
imaging of bland contusions and has been roughly equivalent to CT in imaging hemorrhagic
contusions. Overall, MR has shown approximately 90% of all cortical contusions imaged by
either modality. In general, T2-weighted images are best for evaluating brain contusions. T1-
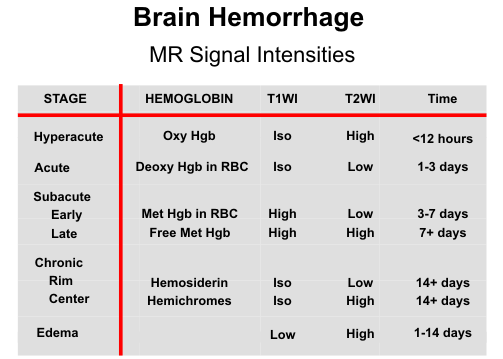
weighted images are helpful to look for any associated hemorrhage. Nonhemorrhagic contusions are hyperintense on T2 and hypointense on T1-weighted scans due to brain edema and increased water content in the lesions. The brain edema increases during the first few days, producing mass effect on adjacent brain structures. With time, the edema subsides and the dead tissues are removed, resulting in areas of encephalomalacia and compensatory focal dilatation of adjacent ventricles and sulci.
The MR appearance of hemorrhagic contusions is more dynamic, changing over time as the internal chemistry of the hematoma changes. In fact, the signal intensities on T1 and T2-weighted images often provide clues about the approximate age of hemorrhagic contusions. The central hypointensity of acute hemorrhagic contusions on T2- weighted images is often highlighted by the surrounding edema. After a few days, methemoglobin forms and gives a mottled pattern of high signal on T1-weighted images owing to the multifocal nature of hemorrhage into cortical contusions.
The brain stem is also subject to injury from head trauma. Although it is protected from direct injury by its location, acceleration/deceleration forces associated with impact to the head may produce displacement and twisting of the brain stem. These forces can result in tearing of penetrating arteries or veins, and compression of the brain stem against the sharp edges of the tentorium or surfaces of the clivus and petrous bones.
SHEAR INJURIES
Severe head injuries are often associated with rotational forces that produce shear stresses on the brain parenchyma. The brain itself has very little rigidity and is extremely incompressible. Brain volume can be decreased only by exerting great pressure. On the other hand, the brain is soft and malleable. Relatively little effort is required to distort the shape of the brain. The parenchyma is of relatively uniform density, except for differences between the CSF of the ventricles and surrounding brain tissue. Slight differences in density also exist between gray and white matter.
When the skull is rapidly rotated, it carries along the superficial brain parenchyma but the
deeper structures lag behind, causing axial stretching, separation and disruption of nerve fiber
tracts. Shear stresses are most marked at junctions between tissues of differing densities. As
a result, shear injuries commonly occur at gray/white matter junctions, but they are also found
in the deeper white matter of the corpus callosum, centrum semiovale, brain stem (mostly the
midbrain and rostral pons) and cerebellum. Lesions in the basal ganglionic regions are usually
found along the borders between the ganglia and the internal or external capsules, in other
words, the deep gray-white matter junctions of the cerebral hemispheres. The thalamic and
basal ganglia injuries are hemorrhagic in slightly more than 50% of cases. On the other hand,
shear injuries of the corpus callosum and centrum semiovale are more often nonhemorrhagic.
Attempts to correlate CT findings with acute and chronic sequelae of closed head trauma
have been discouraging, largely related to the insensitivity of CT to many cerebral injuries.
Chiefly among these, poorly seen by CT and well seen by MR, are the diffuse axonal injuries
or white matter shear injuries. These injuries constitute the most frequent findings on MR in
head trauma, comprising as high as 40% of all lesions. Shear injuries are most often multiple,
ovoid and parallel to white matter fiber bundles. They are hyperintense on T2 and hypointense of T1-weighted scans, unless hemorrhagic components are present, in which case
more complex patterns are observed. During transition phases of hematoma evolution,
combinations of methemoglobin, hemosiderin rings and peripheral edema can result in layers
of differing signal intensity and a target-like appearance. The axial plane is the primary plane
of imaging for both cortical contusions and shear injuries, but supplemental coronal views are
helpful to assess injuries to the body of the corpus callosum and the inferior frontal and
temporal lobes. Fast scan techniques or gradient-echo images have lower resolution but are
useful in uncooperative patients. Contrast enhancement has little role in the evaluation of
brain contusions.
![]()
CHILD ABUSE (Non-accidental trauma)
Unfortunately, there are over a million cases of child abuse in the United States each year. Intracranial injuries may be the result of either direct blows to the head, shaking, whiplash, or strangulation. The presence of an inner hemispheric subdural hematoma is often a tip-off of underlying abuse as it results usually from a whiplash type of injury rather than from a direct blow. There may also be associated subarachnoid hemorrhage as well as subdural hematomas or even subdural hygromas of different ages. One should also be alert for possible underlying non-accidental trauma in the presence of multiple fractures often of different ages. Cerebral anoxia and/or infarction (reversal sign) can occur as a result of strangulation. One should be on the alert for other sites of injury throughout the body.
PENETRATING TRAUMA
The impact cavity of a gunshot wound to the head may vary with the shock wave, the bullet and/or bone fragments. One should try to identify the site of injury, the number of fragments, the location, the path of the wound, and the nature of the hemorrhage. One should also be alert to the path of injury relative to dural sinuses as venous infarctions and/or epidural hematomas may result as well as paranasal sinuses as this can be a source for infections. One should remember that gun shot wounds to the head are not necessarily always fatal.
VASCULAR INJURIES
Traumatic carotid cavernous fistulas can result from an injury to the carotid artery at the level of the cavernous sinus. This causes a high flow arterial venous shunt and often results in painful unilateral pulsating exophthalmos as a result of increased flow in a retrograde direction in the superior ophthalmic vein. This can lead to visual loss. Alternatively, the arterial venous shunt can extend into the inferior petrosal sinus and then into cortical veins of the cerebellum which can lead to subsequent hemorrhage.
Traumatic dissections of the carotid or vertebral arteries in the neck can cause stroke. This can be due to strangulation or whiplash mechanisms which may tear the intima. Typically the carotid is dissected usually just proximal to entering the petrous bone at about the C1-C2 level.
Post-traumatic pseudoaneurysms can occur as a result of vascular injuries intracranially.
These do not have to be located at more typical locations for nontraumatic aneurysms around
the circle of Willis. Vascular spasm can occur as a result of subarachnoid hemorrhage.
![]()
SPECIAL MR TECHNIQUES
Diffusion weighted images (DWI) help elucidate the complex nature of brain injury. First,
several events associated with trauma can result in ischemic brain injury, including cardiac
arrest, hypotension, hypovolemia, hypoxia/anoxia, major vascular laceration, distal embolus,
fat emboli, penetrating vascular injury, and herniation syndromes causing vascular
compromise. Ischemia appears as high signal on DWI and hypointensity on apparent
diffusion coefficient (ADC) maps. Non-ischemic traumatic lesions (contusions, DAI) have
variable diffusion properties. If the cells and axons are completely disrupted, leakage of
intracellular fluid into the extracellular space results in vasogenic edema and high signal
intensity on both DWI and ADC images. On the other hand, minor mechanical damage may
stun cell metabolism and the cell membrane fluid transport system, leading to influx of sodium
and cytotoxic edema. Finally, microvascular arterial injury may produce an element of true
ischemia, whereas microvascular venous injury would lead to vasogenic edema.
![]() ,
,
![]()
Diffusion tensor imaging (DTI) has demonstrated high sensitivity for axonal disruption and is especially helpful for detecting subtle injuries. MR spectroscopy can assess the overall disease burden in traumatic brain injury. Measures of brain NAA levels have been shown to correlate with neuropsychological function.
BIBLIOGRAPHY