IMAGING OF CEREBRAL HEMORRHAGE
AND AV MALFORMATIONS
John R. Hesselink, MD, FACR
INTRACEREBRAL HEMORRHAGE
CT Features

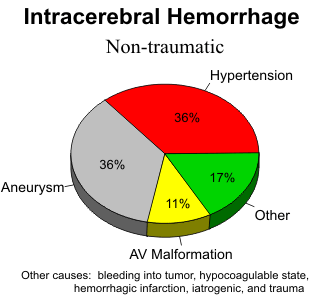
Together, hypertension, aneurysm, and vascular malformations account for 80% of intracerebral hemorrhages. All cerebral hematomas, whatever the cause, have a similar resolution pattern on CT. The rate of resolution depends on the size of the hematoma, usually within one to six weeks, and they resorb from the outside toward the center. Perihematoma low density appears in 24-48 hours. Rim enhancement appears in one week and persists for six weeks. The end result of a hematoma is decreased parenchymal density, focal atrophy and local ventricular dilatation.
MR Appearance
Intracerebral hematomas have a very
dynamic appearance on MR, changing in
signal intensity over time. Acute blood, in the
form the oxyhemogloblin, is isointense with
the brain parenchyma. Within a few hours, the oxyhemoglobin is converted to deoxyhemoglobin
within the hematoma. Deoxyhemoglobin has a predominant effect of shortening T2, resulting in low
signal on T2-weighted images. After three to four days, the deoxyhemoglobin is progressively
converted to methemoglobin, which is a paramagnetic substance. Although methemoglobin shortens
both T1 and T2, the predominant effect is T1 shortening. As a result, at this stage, hematomas are
high signal in both T1-and T2-weighted images. Over the next few months, the methemoglobin is
slowly broken down into hemichromes which produce only mild T1 shortening. Hematomas at this
end stage are slightly high signal on T1-weighted images and remain high signal on the T2-weighted
images. Another interesting phenomenon occurs around the periphery of hematomas. Macrophage
activity results in degradation of the methemoglobin and conversion of the iron moiety to
hemosiderin. Hemosiderin shortens T2 and produces a black ring around the hematoma on
T2-weighted images. We have observed this ring as early as nine days after hemorrhage, and the ring
becomes thicker over time. The amount of hemosiderin varies from one hematoma to another, and
the specific physiologic and chemical factors that influence this are unknown. In small hematomas
(less than 1 cm), we have noted low signal intensity from hemosiderin throughout the cavity. The
length of time that the hemosiderin will remain in the area of a hematoma is also unknown, but we
have observed hemosiderin at the site of a
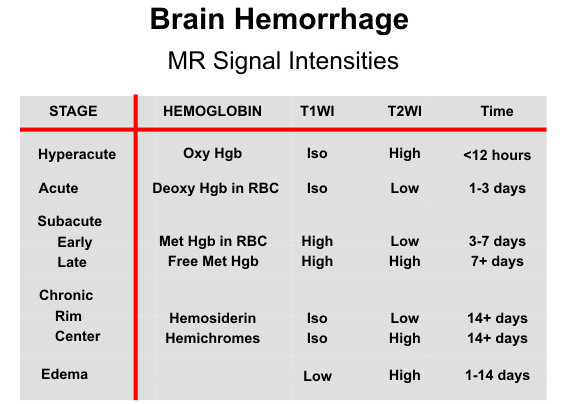
previous hematoma as long as four years following the
primary hemorrhage. From this discussion, it is apparent that the specific signal intensities of a
hematoma on T1- and T2-weighted images provide a clue as to the age of the hemorrhage.
![]()
![]()
Hypertensive Hemorrhage
The criteria for hypertensive hemorrhage include a hypertensive patient, 60 years of age or older, and a basal ganglia or thalamic location of the hemorrhage. A CT scan is the procedure of choice for evaluating these patients. Arteriography is necessary only if one of these criteria is missing. Hypertensive hemorrhages are often large and devastating. Since they are deep hemorrhages and near ventricular surfaces, ventricular rupture is common. One-half of hypertensive hemorrhages occur in the putamen; the thalamus in 25%; pons and brainstem, 10%; cerebellum, 10%, and cerebral hemispheres, 5%.
VASCULAR MALFORMATIONS
Cerebrovascular malformations have been classified into four distinct types including arteriovenous malformation, cavernous angioma, capillary telangiectasia and venous malformation. Capillary telangiectasia is characterized by abnormal, dilated capillaries separated by normal neural tissue. They are usually found in the pons at autopsy and are occult clinically and radiographically. The other three malformations are commonly seen on imaging studies and have distinct features.
Arteriovenous Malformation
The arteriovenous (AV) malformation consists of a congenital abnormality of anomalous, dilated capillaries that result in shunting of blood from the arterial to venous side. AV malformations are by far the most common of the cerebrovascular malfor mations. One-half of patients present with seizures or a neurological deficit due to compression of normal brain or a steal phenomenon. The other half presents with hemorrhage. The hemorrhage is usually more benign than that due to a ruptured aneurysm. Ninety-five percent of AV malformations are in the supratentorial compartment, either in a lobar or deep location and 10% are in the infratentorial region. Dural supply is more commonly found with infra tentorial lesions although it is important to remember than any AV malformation adjacent to a dural surface can receive dural contributions.
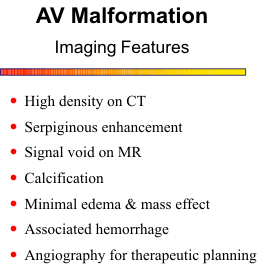
CT features of an AV malformation on plain scan include a high- absorption irregular mass with
large feeding arteries and draining veins, focal areas of calcification and no surrounding edema or
mass effect. The contrast scan shows serpiginous enhancement with prominent arteries and veins.
Due to the rapidly flowing blood from these lesions, a flow void is observed on MR scan. As a result,
the characteristic feeding arteries and draining veins can be imaged without any injection of contrast
material.
![]()
The best MRA sequences for depicting the anatomy of AV malformations are 3D TOF and PC
methods. The TOF technique may not show the draining veins in their entirety due to saturation
effects.
![]() Gd will improve venous visualization, but in general, PC is better for imaging the venous
side. By using two different velocity-encoding factors, 80 and 20 cm/sec, arterial and venous phase
images can be generated. High flow through these lesions often produce turbulence and some signal
loss within the feeding arteries. Selective saturation
pulses can be used to isolate arterial supply.
Gd will improve venous visualization, but in general, PC is better for imaging the venous
side. By using two different velocity-encoding factors, 80 and 20 cm/sec, arterial and venous phase
images can be generated. High flow through these lesions often produce turbulence and some signal
loss within the feeding arteries. Selective saturation
pulses can be used to isolate arterial supply.

As mentioned earlier, flow voids from AV shunting help identify AV malformations on spin-echo MR scans. In fact, spin-echo imaging is probably more sensitive than MRA for detecting AV malformations. Moreover, MRA probably has a limited role in the initial diagnostic workup of AV malformations because a conventional angiogram is required anyway. The proper role for MRA will likely be to follow these lesions, to assess the affects of radiation therapy or embolization procedures, and to check for growth of partially resected lesions.
One should suspect AV malformation as a cause of an intracerebral hemorrhage if the hemorrhage is lobar and away from the territory of the anterior communicating and middle cerebral arteries, and also in deep hemorrhages in younger, normotensive patients. It is important to remember that the hematoma may compress a small AV malformation. If the initial angiogram is negative, a follow-up study should be done one to two months later, after the hematoma and mass effect have resolved. AV malformations can thrombose either spontaneously or due to compression by the hematoma.
Cavernous Angioma

They are characterized by a honeycomb of endothelium-lined vascular spaces, separated by fibrous, collagenous bands with no intervening neural tissue. Most cavernous angiomas are asymptomatic and are noted incidentally on MR scans. They may cause seizures or a focal neurologic deficit, and on occasion they will be of sufficient size to produce symptoms by mass effect. The intralesional hemorrhages are usually small and occult clinically. Multiplicity is common.
Cavernous angiomas invariably contain
hemosiderin from chronic hemorrhage and are
distinctly hypointense on T2-weighted MR images.
Lesion margins are "fuzzy" due to the magnetic
susceptibility effect of the hemosiderin, and a
"blooming effect" occurs with gradient-echo
sequences. Calcification is often present. Mild
enhancement can be obscured by the hemosiderin.
![]()
Larger cavernous angiomas have a more complex
appearance from multiple hemorrhages of varying
ages. Hemosiderin lines the perimeter of these lesions
and also outlines the internal compartments that
contain various components of hemorrhage.
![]()
Venous Malformation
Venous malformations involve only venous structures and are usually incidental findings on MR,
CT or arteriography. Venous malformations are less likely to bleed than the arteriovenous variety.
Those that are located in the cerebellum or brainstem seem to be at a higher risk for bleeding than the
supratentorial ones. Also, cavernous angiomas have been shown to be associated with some venous
angiomas, which may increase the risk of hemorrhage.
![]()
Venous malformations have a characteristic appearance on MR and contrast-enhanced CT scans.
A radial pattern of small veins join to form a single large vein that drains toward the cortical surface.
Drainage into the deep venous system is uncommon. Usually, flow is sufficiently fast to produce a
flow void on spin-echo MR images. They are also routinely visualized with MR angiography.
![]()
REFERENCES