DEMYELINATING DISEASES OF THE BRAIN
John R. Hesselink, MD, FACR
MR imaging is exquisitely sensitive for detecting brain abnormalities. Particularly in the evaluation of white matter diseases, MR far outperforms any other imaging technique. Lesions that may be quite subtle or even invisible on CT are often clearly seen on the MR scan. The MR signal characteristics of white matter lesions are similar and relatively nonspecific, but other distinguishing features are often present to assist in diagnosis, such as the pattern of the abnormality, location, and enhancement features.
The white matter is affected by many disease processes. The primary demyelinating disease is multiple sclerosis, but many other metabolic and inflammatory disorders result in deficient or abnormal myelination. Histologically, myelin abnormalities are either demyelin-ating or dysmyelinating. Demyelination implies destruction of myelin. Dysmyelination refers to defective formation or maintenance of myelin resulting from dysfunction of the oligodendrocytes. Most of the dysmyelinating disorders are caused by metabolic defects that present in infancy. White matter diseases in older children and adults are generally demyelinating or a combination of the two processes.
NORMAL WHITE MATTER
The white matter of the brain is located in the central and subcortical regions of the
cerebral and cerebellar hemispheres and accounts for about 60 % of the total brain volume.
The white matter includes the major commissural tracts, the cortical association fibers, and
all the cortical afferent and efferent fibers. Histologically, the white matter contains nerve
fibers, supporting cells, interstitial space, and vascular structures. White matter consists
mostly of axons with their envelope of myelin, along with two types of neuroglia: oligo-dendrocytes and astrocytes. Axons are extensions of neurons that reside within the gray
matter of the brain, spinal cord, and ganglia. The myelin is produced and maintained by oligodendrocytes. Myelin functions as an insulator of the axons, and its structure facilitates rapid
transmission of impulses.
![]()
Myelin has relatively short T2 and T1 relaxation times, primarily owing to its lipid content. As a result, normal myelin is hypointense to gray matter on T2-weighted images and hyperintense on T1-weighted images. If a disease process reduces the myelin content, the white matter becomes less hydrophobic and takes on more water. Less myelin and more water protons prolong the relaxation times of both T1 and T2, resulting in more signal on T2-weighted and less signal on T1-weighted images.
AGING EFFECTS ON THE WHITE MATTER
As a result of the high sensitivity of T2-weighted spin-echo pulse sequences, MR images
frequently reveal high-signal foci within the subcortical white matter. Estimates of the
incidence of these hyperintensities in the brains of healthy, elderly persons have ranged from
30 to 90 per cent.
![]() ,
,
![]() ,
,
![]() Gerard and Weisberg3 found subcortical lesions in only 10 per cent of
patients older than 60 years unless cerebrovascular symptoms or risk factors were present;
if both were present, the incidence increased to 84 per cent. To a certain extent, the presence
of these hyperintensities limits the sensitivity of MR for white matter disease. They are often
normal variants or related to deep white matter ischemia, but they can be mistaken for, or can
obscure more serious pathology.
Gerard and Weisberg3 found subcortical lesions in only 10 per cent of
patients older than 60 years unless cerebrovascular symptoms or risk factors were present;
if both were present, the incidence increased to 84 per cent. To a certain extent, the presence
of these hyperintensities limits the sensitivity of MR for white matter disease. They are often
normal variants or related to deep white matter ischemia, but they can be mistaken for, or can
obscure more serious pathology.
Virchow-Robin Perivascular Spaces
When nutrient vessels penetrate the brain substance, the pia mater is carried along with the vessel down to the capillary level. The small subarachnoid space that follows the pia is called the Virchow-Robin (VR) space. These perivascular CSF spaces appear as punctate areas of high signal on T2-weighted images.
Occasionally, very large (1 cm or more) VR spaces will be observed in the basal ganglia region. If the patient is young and has no risk factors for vascular or degenerative disease, these large VR spaces are probably normal variants, representing a confluence of penetrating arteries and veins.
Not infrequently, VR spaces in the brainstem will be sufficiently large to be seen on MR images. On T2-weighted axial images they are sectioned longitudinally and appear as hyperintense linear structures coursing in a ventrolateral direction. Their linear character generally distinguishes them from small brainstem infarcts.
If the brain become atrophic and loses volume, it retracts away from the vessels and
extracellular fluid fills the space. On postmortem studies, these perivascular fluid spaces
appear like a network of tunnels within the brain substance. These changes have been termed
état criblé (sieve-like). These fluid spaces simply represent dilated VR spaces. Brain atrophy
results in dilated VR spaces in the same manner that the cortical sulci become enlarged. As
expected, in older patients with atrophic brains the VR spaces are larger and appear more
numerous.
![]()
Deep White Matter Ischemia
Pathophysiology
As the brain ages, structural, chemical, and metabolic changes occur that are reflected on
the MR images. Most important for MR interpretation are the changes related to deep white
matter ischemia. The deep white matter of the cerebral hemispheres receives its blood supply
from long, small-caliber arteries and arterioles that penetrate the cerebral cortex and traverse
the superficial white matter fiber tracts. The white matter does not have as generous a blood
supply as the gray matter and is more susceptible to ischemia. As the nutrient arteries become
narrowed by arteriosclerosis and lipohyaline deposits within the vessel walls, the white matter
becomes ischemic on a chronic basis. Pathologically, one of the first changes in the aging
brain is an increase in the perivascular interstitial fluid, predominantly at the arteriolar level
of the vascular tree. With continued progressive ischemia of the white matter, additional
histologic changes are observed, including atrophy of axons and myelin and tortuous,
sclerotic, and thickened vessels. Maintenance of the myelin becomes deficient, resulting in
"myelin pallor" on microscopic sections. Mild gliosis and increased interstitial fluid
accompany the changes in the myelin. Necrosis is not seen until severe ischemia leads to
frank infarction of brain tissue.
![]()
As mentioned previously, the observed changes are secondary to chronic ischemia of the deep white matter and are found more often in patients with ischemic cerebrovascular disease, hypertension, and aging. A combination of arteriolar disease and episodic brain hypoperfusion probably leads to the histopathologic changes. Hypoperfusion can be caused by episodes of hypotension, hypoxia secondary to cardiac or carotid artery disease, hypertension, and/or aging.

The most common locations for the hyperintensities are the subcortical and periventricular white matter, optic radiations, basal ganglia and brain stem, in decreasing order of frequency. The lesions are hyperintense on T2, proton density, and FLAIR images and have well defined but irregular margins. They are mildly hypointense on T1 weighted images, but the contrast between the lesions and the normal brain is much less than that on T2 weighted images. The white matter changes do not exhibit restriction diffusion, are not associated with breakdown of the blood brain barrier and do not enhance. The process tends to be multifocal, but as the lesions enlarge, a more confluent pattern may develop.
Another pattern of white matter abnormality associated with deep white matter ischemia
is a more or less continuous band of abnormal signal bordering the lateral ventricles.
Normally, interstitial fluid does not accumulate around the ventricles except for small "CSF
caps" around the frontal and occipital horns. White matter ischemia results in mild interstitial
edema that exceeds the capacity of the ependymal transport system. The chronic
accumulation of water around the ventricles adversely affects myelin maintenance and over
time induces partial but irreversible loss of myelin that is reflected as myelin pallor of the
periventricular white matter on pathologic examination. On T2-weighted MR images this
white matter abnormality appears as a hyperintense band of variable thickness located along
the dorsolateral angles of the ventricles, closely apposed to the ependymal surface. A slightly
heterogeneous texture and irregular outer margins distinguish it from the phenomenon of
transependymal CSF flow or "periventricular halo" of obstructive hydrocephalus (see
"Differential Diagnosis" further on). On T1-
weighted images the periventricular lesions
are lower signal than the subcortical white
matter lesions, probably due to increased
water content.
![]()
MULTIPLE SCLEROSIS
On histologic examination, acute MS
plaques show partial or complete
destruction and loss of myelin with sparing
of axon cylinders. They occur in a
perivenular distribution and are associated
with a neuroglial reaction and infiltration of mononuclear cells and lymphocytes. The
perivascular demyelination gives the appearance of a finger pointing along the axis of the
vessel. In the pathologic literature these elongated lesions have been named "Dawson's
fingers." Active demyelination is accompanied by transient breakdown of the blood-brain
barrier. Chronic lesions show predominantly gliosis. MS plaques are distributed throughout
the white matter of the optic nerves, chiasm and tracts, the cerebrum, the brain stem, the
cerebellum and the spinal cord.
![]() ,
,
![]()
Imaging Features
MS plaques are hyperintense on T2-weighted and FLAIR images and hypointense on T1-weighted scans. Specific signal intensities of MS lesions will vary depending on the magnetic field strength, the pulse sequence parameters, and partial volume effects. Occasionally, acute plaques may have a thin rim of relative T2 hypointensity or T1 hyperintensity. The T1 hyperintensity is attributed to free radicals, lipid-laden macrophages, and protein accumulations.
MS plaques are usually discrete foci with well-defined margins. Most are small and irregular, but larger lesions can coalesce to form a confluent pattern. Multiple focal periventricular lesions can give a "lumpy-bumpy" appearance to the ventricular margins. As a result of their perivenular distribution, many periventricular plaques have an ovoid configuration, with their long axis oriented transversely on an axial scan. The ovoid lesion is the imaging correlate of "Dawson's finger." In general, MS plaques have a homogeneous texture without evidence of cystic or necrotic components. Hemorrhage is not a feature of MS lesions. Edema and mass effect are also uncommon.
The periventricular white matter is a favorite site for MS plaques, particularly along the
lateral aspects of the atria and occipital horns. The corpus callosum, corona radiata, internal
capsule, visual pathways, and centrum semiovale are also commonly involved. When more
than a few lesions are present, symmetric involvement of the cerebral hemispheres seems to
be the rule. Any structures that contain myelin can harbor MS plaques, including the brain
stem, spinal cord, subcortical U-fibers, and even within the gray matter of the cerebral cortex
and basal ganglia. A distinctive site in the brain stem is the ventrolateral aspect of the pons
at the fifth nerve root entry zone.
![]() Brain stem and cerebellar plaques are more prevalent in
the adolescent age group.
Brain stem and cerebellar plaques are more prevalent in
the adolescent age group.
![]()
Lesions of the corpus callosum have been a special focus of study. On axial sections, plaques in the corpus callosum above the lateral ventricles have a transverse orientation along the course of the nerve fiber tracts and vessels. Sagittal FLAIR images are especially helpful to depict the small callosal lesions closely apposed to the superior ependymal surface of the lateral ventricles. Early edema and demyelination along subependymal veins produce a striated appearance. Atrophy of the corpus callosum is common in long-standing, chronic MS and is seen best on T1-weighted sagittal images.
Involvement of the visual pathways, particularly the optic nerves, frequently occurs
sometime during the course of disease. Patients may present with optic neuritis, although in
about half of those cases, MRI will unveil other silent lesions in the brain. Imaging plaques
in the optic nerves is a challenge even for MRI. Unenhanced spin-echo sequences are not
very sensitive, and generally some type of fat suppression is required. Probably the most
sensitive method for detecting acute MS of the optic nerves is the combination of gadolinium
enhancement and fat suppression.
![]()
The spinal cord is commonly involved by MS, and patients may present with a transverse
myelitis. All levels of the cord can be affected, but most plaques are found in the cervical
region. Since the white matter fiber tracts are positioned along the outer aspects of the cord,
MS plaques are often based along a pial surface and have an elongated configuration. Signal
characteristics are similar to lesions in the brain. Edema associated with acute plaques may
lead to cord swelling, simulating an intramedullary tumor. In chronic MS, cord atrophy can
result from focal lesions or axonal degeneration from distal disease.
![]()
Nonenhanced MR cannot judge lesion activity, because plaques almost always remain
evident after the acute clinical episode. Although the water content of acute plaques
decreases over time, the T1 and T2 relaxation times of acute and chronic plaques have
sufficient overlap that quantitative MR cannot distinguish between old and new lesions.
Quantitative brain analyses of MS patients have shown that the T1 and T2 relaxation times are
prolonged not only in acute and chronic plaques but also in normal-appearing white matter.
![]() Occasionally, a “dirty white matter” appearance can be seen on T2-weighted images. Diffuse
white matter involvement has been confirmed further with magnetization transfer (MT)
measurements.
Occasionally, a “dirty white matter” appearance can be seen on T2-weighted images. Diffuse
white matter involvement has been confirmed further with magnetization transfer (MT)
measurements.
![]()
Gadolinium enhancement
Since acute MS plaques are associated with transient breakdown of the blood-brain
barrier, gadolinium contrast agents will produce enhancement of these lesions on T1-weighted
images. Enhancement will be observed for 8 to 12 weeks following acute demyelination.
Thus, Gd-enhanced MR can be used to assess lesion activity just like contrast-enhanced CT.
Either nodular or ringlike enhancement may be seen early after contrast injection, but the
central areas tend to fill in and become more homogeneous on delayed scans. Immediate
postcontrast scans are most sensitive for detecting MS, and delayed scanning is not necessary.
Contrast-enhanced MR can be used to follow the progression of disease and to assess the
response to therapy.
![]()
Occasionally, large plaques, also called tumefactive MS, may produce mass effect and
simulate other mass lesions. However, compared with neoplastic or inflammatory processes,
MS plaques have minimal surrounding edema and relatively less mass effect for the overall
size of the white matter lesions. Balo's concentric sclerosis has a unique MR appearance.
Like tumefactive MS, the plaques usually are quite large, but in addition, a concentric
laminated pattern is seen on T2 and T1-weighted images. Similarly, post-contrast images
often show rings of enhancement alternating with non-enhancing regions during the acute
phase.
![]()
INFECTIOUS AND INFLAMMATORY DISORDERS
Progressive Multifocal Leukoencephalopathy
PML has an affinity for subcortical white matter, and the classic distribution is in the
parietal-occipital lobes. It is not primarily a periventricular process, but as the disease
progresses, the deeper white matter is also affected. Any white matter structures can be
involved, but lesions of the corpus callosum are much less common than in multiple sclerosis,
for example. Brain stem and cerebellar lesions are found in about one-third of patients;
occasionally, they can be the solitary presenting lesion. Basal ganglia and thalamic sites
generally represent extension from lesions in the internal capsule or damage to white matter
fibers coursing through the gray matter structures.
![]()
The white matter lesions of PML are patchy and round or oval at first, but then become confluent and large. The process is often distinctly asymmetric and initially involves the peripheral white matter, following the contours of the gray-white matter interface to give outer scalloped margins. Lesions tend to be homogeneous with well-defined margins. The prolonged T1 and T2 relaxation times reflect the loss of myelin and increased water. Mass effect and contrast enhancement are rarely seen.
Post-Infectious Encephalitis
Post-infectious encephalitis, also called acute disseminated encephalomyelitis (ADEM),
is a demyelinating disease that is thought to be of autoimmune origin. It usually occurs within
two weeks after one of the childhood viral infections, such as measles or chicken pox, or
following vaccination against rabies or smallpox. It has also been reported in association with
chronic Epstein-Barr virus infection. The clinical picture is one of abrupt onset with a
monophasic course, to distinguish it from MS and most other white matter diseases. Initial
headache, seizures, and drowsiness may progress to profound lethargy and even coma. Brain
stem involvement can produce nystagmus, diplopia, and dysarthria. Since it is a
myelinoclastic process, lesions usually correlate with discrete clinical symptoms.
![]() Although
variable, recovery is often surprisingly good. Acute hemorrhagic leukoencephalitis is a more
fulminant variant of ADEM.
Although
variable, recovery is often surprisingly good. Acute hemorrhagic leukoencephalitis is a more
fulminant variant of ADEM.
Lesions are found in the white matter of the brain stem, cerebrum, and cerebellum, and as a rule are asymmetric and few in number. There is no predilection for the periventricular white matter. The lesions are typically hyperintense on T2 and hypointense on T1-weighted images and without hemorrhage or calcification. Despite the acute demyelinating picture, contrast enhancement is usually mild and spotty in nature.
HIV Encephalitis
A subacute encephalitis involving the white matter is seen in up to 30% of patients with
AIDS related to direct invasion of neurons by the neurotropic retrovirus, human
immunodeficiency virus type 1 (HIV). Presenting symptoms include headache, memory loss,
language difficulty, movement disorders, and various sensory deficits. More advanced disease
is signaled by behavioral disorders, loss of bowel and bladder control, and the AIDS dementia
complex. HIV and cytomegalovirus often coexist in brain specimens taken from patients with
AIDS; however, HIV seems to be the predominant etiology for the microglial nodules and
multinucleate giant cells, the pathologic hallmarks of the white matter encephalitis.
![]() ,
,
![]()
The MR picture is one of bilateral, diffuse, patchy to confluent areas of increased signal
intensity on T2-weighted images with poorly defined margins involving the white matter of
the cerebrum, cerebellum and brain stem. The white matter changes are not as striking as in
many other demyelinating diseases. Initially, the images may show only a hazy mottled
pattern of hyperintensity within the centrum semiovale. HIV encephalitis does not enhance
with gadolinium. The MR appearance is distinct from that of PML, and the clinical setting
readily separates it from other white matter abnormalities. As with PML, the white matter
abnormality on MRI often improves or remains stable after therapy with protease inhibitors,
correlating with clinical improvement.
![]()
RADIATION INJURY
Radiation injury to the brain has a progressive and insidious course. The effects on the
brain can be focal or diffuse, depending on whether whole brain or more focused radiation
was given. The clinical picture is variable. Many patients are entirely asymptomatic, and as
a rule, severe imaging abnormalities are required in order for patients to have symptoms.
Impairment of mental function is the most common problem, and may include personality
change, memory deficiencies, confusion, learning difficulties, and in severe cases, dementia.
![]()
A few months following radiation, demyelination is seen histologically, associated with proliferation of the glial elements and mononuclear cells. This condition can progress to irreversible damage to the capillary endothelium, perivascular inflammation, diffuse vasogenic edema of the cerebral white matter, necrotic foci, vacuolation, and petechial hemorrhage. Endothelial hyperplasia also occurs, resulting in reduced cerebral blood flow. The pathologic changes of radiation necrosis continue to evolve for a number of years after the initial radiation. The location and amount of brain injury are related to the radiation dose, fractionation methods, and the portals used.

The effects of late radiation injury to the brain are first detected on imaging studies about 6 to 8 months following the initial therapy. MR detects more lesions than CT and the abnormalities appear more extensive on MR than on CT. The characteristic pattern of diffuse radiation injury is symmetric, high-signal foci on T2-weighted images in the periventricular white matter. There is initial sparing of the corpus callosum and the subcortical arcuate fibers. The changes parallel those seen in ischemia, but the lesions are more prevalent and a confluent pattern usually develops. As the process extends outward to involve the peripheral arcuate fibers of the white matter, the margins become scalloped, a helpful feature in the differential diagnosis. With time, atrophy becomes a part of the picture, as shown by enlargement of the ventricles and cortical sulci. There is relative sparing of the posterior fossa, basal ganglia, and internal capsules, but in more severe cases these structures are also involved. Deposition of hemosiderin in the basal ganglia has been reported. As observed histologically, imaging findings may continue to progress for 2 or more years after radiation therapy.
With focal high-dose therapy, radiation necrosis may lead to profound edema, mass effect,
and contrast enhancement with gadolinium. Especially in these cases, distinguishing radiation
change from recurrent tumor can be extremely difficult, if not impossible. Frequently, biopsy
is required to clarify the issue.
![]()
ACQUIRED TOXIC-METABOLIC DISORDERS
Effects of Chemotherapeutic Agents
Many of the cancer chemotherapeutic drugs are neurotoxic. The blood-brain barrier (BBB) limits the direct entry of these substances into the brain, but the BBB may be damaged by the disease process or by the therapeutic agent itself. Also, in the case of CNS disease, agents may be given to open the BBB to allow greater access of the anticancer drug to the tumor. Lipid solubility and intrathecal administration are other factors that increase delivery to the brain. The chemotherapeutic agents commonly associated with leukoencephalopathy include methotrexate, cis-platin, cytosine arabinoside (ARA-C), carmustine (BCNU), and thiotepa.

Both acute and delayed effects are observed with chemotherapy. The acute changes develop during therapy, often within the first few days of initiating treatment. High-dose intravenous methotrexate is the most common cause. The bilateral diffuse white matter hyperintensity is transient in nature, and patients may be entirely asymptomatic.
The delayed effects of chemotherapy
range from asymptomatic white matter
hyperintensities to a severe necrotizing
leukoencephalopathy. The onset of
clinical and imaging findings is earlier than that observed with radiation, usually a few weeks
or months following therapy. The reported incidence of necrotizing leukoencephalopathy
varies widely, but is much higher with CNS leukemia, or when combinations of intravenous
and intrathecal chemotherapy and radiation is employed.
![]()
MR imaging initially reveals patchy involvement of the periventricular white matter and centrum semiovale, which over time evolves to a confluent pattern. The process tends to spare the deep white matter tracts, brain stem, and cerebellum. Enhancement or mass effect are seen only in the most severe cases. Long term follow-up often shows some brain atrophy, and in children treated for cancer, cerebral calcification is commonly found.
Posterior Reversible Encephalopathy Syndrome
The posterior reversible encephalopathy syndrome (PRES) describes the combination of
posterior cerebral involvement and brain dysfunction that occurs with this entity. Patients may
present with headache, confusion, visual disturbance, and seizures. Several diseases and
clinical conditions are associated with PRES, but the root cause seems to be acute or
subacutely elevated blood pressure. Hypertension can occur with drug ingestion, chronic renal
disease, autoimmune nephritis (systemic lupus erythematosus), Wegener's granulomatosis,
acute renovascular hypertension, pheochromocytoma, preeclampsia and eclampsia, and
immunosuppressant therapy.
![]()
MRI reveals lesions with high signal intensity on FLAIR and T2 weighted nonenhanced
images. There is a predilection for the posterior cerebral white matter, and in severe cases
the adjacent gray matter can be involved. Extension into the frontal lobes is not uncommon,
but isolated anterior white matter involvement is unusual. Contrast enhancement is patchy if
present at all. The white matter lesions of PRES do not exhibit restricted diffusion.
![]() The
white matter injury is largely reversible. At least, with correction of the hypertension, the MRI
findings resolve within a few weeks and follow clinical recovery.
The
white matter injury is largely reversible. At least, with correction of the hypertension, the MRI
findings resolve within a few weeks and follow clinical recovery.

Several immunosuppressive agents have been associated with PRES. The cyclosporins
are the most widely used, and neurological complications occur in about 20 per cent of
transplant patients receiving these agents. Cyclosporine has been shown to induce endothelial
release of vasoactive mediators. Hypertension is present in 90% of patients, especially when
combined with steroids. Although cyclo-
sporine may have some direct neuro-toxicity
on the myelin, symptoms and imaging are like
those of hypertensive encephalopathy.
![]() White
matter damage has also been reported with
tacrolimus (FK 506), another commonly used
immunosuppressant drug. Sudden changes in
electrolyte or fluid balance can occur due to
diarrhea, poly- or oliguria.
White
matter damage has also been reported with
tacrolimus (FK 506), another commonly used
immunosuppressant drug. Sudden changes in
electrolyte or fluid balance can occur due to
diarrhea, poly- or oliguria.
![]()
Central Pontine Myelinolysis
Central pontine myelinolysis, also called
osmotic demyelination syndrome, is a disorder characterized pathologically by dissolution of
the myelin sheaths of fibers within the central aspect of the basis pontis. In extreme cases
there may be extension to the pontine tegmentum, midbrain, thalamus, internal capsule, and
cerebral cortex. The myelinolysis occurs with relative sparing of the nerve cells and axon
cylinders. Many patients are asymptomatic, and at the other extreme are patients whose
symptoms are masked by coma. Most clinically diagnosed cases present with spastic
quadriparesis, pseudobulbar palsy, and acute changes in mental status, with progression
possible to altered levels of consciousness and death. Survival is possible with varying residual
neurologic deficits. Although initial reports were largely confined to chronic alcoholics,
central pontine myelinolysis has also been seen in patients with electrolyte disturbances,
particularly hyponatremia that has been rapidly corrected, and in liver transplant patients being
immune-suppressed with cyclosporine.
![]()
The lesions on MR are seen best as areas of hypointensity on IR images and hyperintensity
on T2-weighted images in the central pons with sparing of the pontine tegmentum and
ventrolateral pons. Lesions have an oval shape on sagittal images, a bat-wing configuration
on coronal images and various shapes on the axial images.
![]() The extrapontine lesions often
resolve completely, leaving some residual pontine abnormality. Enhancement is not a feature
of this disease, but severe cases may show peripheral enhancement of the pontine lesions.
Diffusion techniques usually show increased diffusibility and increased apparent diffusion
coefficient (ADC), but restricted diffusion and decreased ADC can be seen acutely during the
first week.
The extrapontine lesions often
resolve completely, leaving some residual pontine abnormality. Enhancement is not a feature
of this disease, but severe cases may show peripheral enhancement of the pontine lesions.
Diffusion techniques usually show increased diffusibility and increased apparent diffusion
coefficient (ADC), but restricted diffusion and decreased ADC can be seen acutely during the
first week.
HEREDITARY METABOLIC DISORDERS
This group includes a long list of diseases that affect the gray and white matter to varying degrees. This section discusses briefly the major diseases that affect primarily the white matter, including the classic leukodystrophies. The names and terminologies of these disorders are confusing because they were derived from the pathologic literature before their metabolic defects were discovered. As the specific biochemical and enzyme defects are being elucidated, these diseases are being classified more appropriately.
The classic leukodystrophies include adrenoleukodystrophy, Krabbe's globoid cell, and
metachromatic leukodystrophy, and a few other less well known entities. They have in
common a genetic origin and involve the peripheral nerves as well as the central nervous
system. Each is caused by a specific inherited biochemical defect in the metabolism of myelin
proteolipids that results in abnormal accumulation of a metabolite in brain tissue. Progressive
visual failure, mental deterioration, and spastic paralysis develop early in life, however,
variants of these diseases have a more delayed onset and a less progressive course. The other
primary white matter disorders include Alexander's disease, Canavan disease, Cockayne's
syndrome, and Pelizaeus-Merzbacher's disease.
![]()
All of the above white matter diseases are characterized by symmetric massive
involvement of the white matter. MR imaging is very sensitive for detecting the white matter
damage, but it is not very specific. Adrenoleukodystrophy primarily involves the parieto-occipital and temporo-occipital lobes but extends forward as the disease progresses. Sites of
active demyelination along the advancing edges may be associated with BBB disruption and
enhance with contrast. Unlike the focal plaque-like character of MS, adrenoleukodystrophy
tends to be contiguous within fiber tracts and often is confluent within the larger white matter
bundles of the centrum semiovale.
![]() Alexander's disease has a propensity to involve the
frontal lobes.
Alexander's disease has a propensity to involve the
frontal lobes.
Two metabolic disorders, Hurler's disease and Lowe's syndrome, are associated with cystic changes in the cerebral white matter.
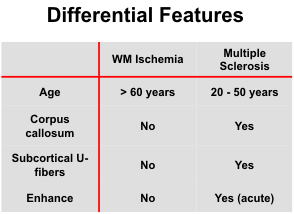
DIFFERENTIAL DIAGNOSIS
Since the periventricular hyperintensities have common histologic features, such as increased interstitial water, demyelination, and gliosis, it should not be surprising that their MR appearance can be quite similar. Nevertheless, differential clues are often present on the images. The normal Virchow-Robin spaces should not be a problem. They are round, are no more than 1 or 2 mm in size, and are seen on the higher axial sections through the cerebral hemispheres and on lower sections through the basal ganglia at the level of the anterior commissure.
Enhancement is a very helpful feature. Acute MS plaques, subacute infarcts, radiation necrosis, and adrenoleukodystrophy are associated with breakdown of the blood-brain barrier and enhance with gadolinium. Enhancement patterns provide additional diagnostic clues. The CSF spaces and chronic changes of white matter ischemia and aging will not enhance. Most of the hereditary metabolic disorders and the immune mediated inflammatory disorders do not enhance.
Many of the other white matter diseases have special features. PML occurs in an immunocompromised host and involves the peripheral white matter in a patchy and asymmetric fashion. The hereditary metabolic disorders occur in children and exhibit a symmetric, diffuse and confluent pattern of involvement. Other infectious and inflammatory disorders have an acute clinical course and a history of a recent viral infection, vaccination, or AIDS.
Cerebral arteritis, secondary to collagen vascular disease or granulomatous disease, can
also result in multifocal periventricular hyperintensities. Moreover, these diseases occur in
young adults and can produce a neurologic picture similar to that of MS. Associated systemic
features are important diagnostic clues for vasculitis, and the brain images usually reveal
cortical infarcts in addition to the periventricular lesions.
![]()
Migraine is another neurologic problem associated with periventricular abnormalities.
In a study by Soges and colleagues
![]() periventricular hyperintensities were found in 41 per cent
of patients with classic or common migraine and in 57 per cent of patients with complicated
migraine. The white matter lesions resemble deep white matter ischemia or vasculitis more
than multiple sclerosis, and the classic pattern of headaches usually identifies these patients.
periventricular hyperintensities were found in 41 per cent
of patients with classic or common migraine and in 57 per cent of patients with complicated
migraine. The white matter lesions resemble deep white matter ischemia or vasculitis more
than multiple sclerosis, and the classic pattern of headaches usually identifies these patients.
Periventricular leukomalacia is caused by neonatal hypoxia or ischemia, but it may be
imaged during adulthood when these patients are being re-evaluated or seen for other
neurological problems. It affects predominantly the periventricular white matter along the
posterior bodies and atria of the lateral ventricles. The involvement tends to be symmetrical
and is often associated with regional loss of white matter volume and ventricular dilatation.
The lesions have scalloped outer margins, and the ventricular surface may also be irregular
due to coalescence of cystic components with the adjacent ventricles.
![]()
A number of processes can cause a more or less continuous band of abnormal signal
within the white matter bordering the ventricles. Since the abnormal signal is due to fluid
accumulation and some loss of myelin, it is hyperintense on T2-weighted scans and
hypointense on T1-weighted scans. Transependymal CSF flow from hydrocephalus usually
appears as a smooth halo of relatively even thickness. It also has the associated findings of
lateral ventricles enlarged out of proportion to the cortical sulci, dilated inferior recesses of
the third ventricle, and smooth elevation and thinning of the corpus callosum. As mentioned
earlier, deep white matter ischemia can result in myelin pallor of the periventricular white
matter, but it is more heterogeneous, the margins are less sharply defined, and it involves
predominantly the white matter along the upper outer margins of the lateral ventricles. CMV
ventriculitis or ependymitis results from reactivation of a latent herpesvirus in an immune-compromised host, usually in the setting of HIV infection. T2 hyperintensity often outlines
the entire lateral and third ventricles, and in severe cases the fourth ventricle as well. The
associated inflammatory reaction results in gadolinium enhancement of the ependyma and
subependymal white matter. The enhancement is linear, continuous, and of variable
thickness.
![]()
All of the above features are helpful in evaluating periventricular white matter abnormalities. Sometimes, a definitive diagnosis cannot be made from the images alone, and a list of differential diagnoses must be considered and correlated with clinical information. Occasionally, one or two punctate subcortical hyperintensities may be found in a young healthy patient with no focal symptoms, no risk factors for vascular diseases, no prior history of trauma or neurologic disease, and imaging features that do not fit with any of the above entities. Correlative pathology is not available in these cases, but the lesions most likely represent focal gliosis related to a prior subclinical infection (prenatal or postnatal) or some indeterminate developmental event. None of these punctate lesions have ever been reported to evolve into something significant, such as tumor, or to be a seizure focus, and therefore, in all probability they are of no clinical significance.
REFERENCES