IMAGING THE SPINAL CORD AND INTRADURAL DISEASE
John R. Hesselink, MD, FACR
Cross-sectional imaging of the spine and spinal cord has contributed substantially to the detection
and characterization of spinal diseases. MR has several advantages over CT and has largely replaced
CT as the primary imaging modality. A major benefit of MR is the ability to directly image the spinal
cord. The MR images not only show the external contours of the cord but also reveal the internal
texture of normal and diseased segments. Moreover, since different signal intensities are obtained
from the cortical bone, epidural fat, cerebral spinal fluid (CSF), and spinal cord, MR is well-suited
for imaging extradural, subarachnoid, and intramedullary abnormalities within the spinal canal.
![]()
EXAMINATION TECHNIQUE
In the diagnostic work-up of spinal disease, multiple anatomic sites need to be imaged, including
the intervertebral disk, spinal canal, spinal cord, nerve roots, neuroforamina, facet joints, and the soft
tissues within and surrounding the spine. Many pulse sequences are available, and specific protocols
vary among different MR sites. There is general agreement that the spine needs to be imaged in at
least two planes, and surface coils are used almost exclusively. In the cervical region a T2-weighted
sequence is mandatory to assess the spinal cord. Most protocols include a T1-weighted sequence and
some type of T2-weighted sequence to give a myelographic effect.
![]() Fast spin-echo (FSE) techniques
allow enormous time savings, and if available, they have replaced conventional spin-echo for T2-weighted imaging of the spine.
Fast spin-echo (FSE) techniques
allow enormous time savings, and if available, they have replaced conventional spin-echo for T2-weighted imaging of the spine.
Gadolinium injection with T1-weighted imaging is essential to evaluate enhancing lesions. Fat-suppression is helpful to eliminate competing fat signal from bone marrow and other soft tissues.

SPINAL CORD
Intramedullary Tumors
Expansion of the cord is the hallmark of an
intramedullary neoplasm. Most cord tumors are
hyperintense on T2-weighted images and hypo
intense on T1-weighted images. Areas of
increased T2 signal extending above or below
the expanded cord segment represent micro
infiltration of tumor or associated edema.
Conventional MR techniques cannot reliably
make this distinction. Gadolinium enhances
areas of blood-brain barrier breakdown to help
detect, characterize and define the extent of
intramedullary neoplasms.
![]()
Focal areas of hemorrhage, intratumoral
cysts or necrotic cavities can give cord tumors a
heterogeneous appearance on MR scans. The
signal intensity depends on the concentration of protein within the cysts and the specific components
of hemorrhage that are present.
![]()
In general, the MR appearance of cord neoplasms varies widely, and making a specific diagnosis
is difficult in most cases. An enhancing tumor in the conus is highly likely to be an ependymoma.
In the cervical and upper thoracic cord, the frequency of ependymoma and astrocytoma is about
equal. The classic features of hemangioblastoma include a cyst with an enhancing mural nodule. Fat
signal components are an important clue for a lipoma or teratoma. Hematogenous metastases from
breast or lung carcinoma and melanoma may produce a similar appearance to primary intramedullary
neoplasms, although metastases may involve several segments of the cord and subarachnoid spread
of tumor is common. Primary CNS lymphoma can, on occasion, involve the spinal cord.
![]()
Syringohydromyelia
Syringomyelia refers to a cavity in the spinal cord extending lateral to or independent of the central canal. Hydromyelia refers to a dilated central canal of the cord. Most cavities involve both parenchyma and central canal, and the term syringohydromyelia reflects this combined phenomenon. The clinical presentation is variable depending on the location and extent of the syrinx. The classic picture is segmental weakness and atrophy of the hands and arms, with loss of reflexes and segmental anesthesia.

In a large series reported by Sherman et al,
![]() 41% had syringomyelia associated with tonsillar
ectopia (Chiari I malformation), 28% had posttraumatic syrinx, 15% were associated with neoplasm,
and 15% were idiopathic. At least one third of patients with nonneoplastic cavities had associated
high-signal intensity in the cord contiguous to the cavity or in the parenchyma distal or proximal to
the cavity. This high-signal intensity in a simple
syrinx is speculated to represent gliosis, edema,
demyelination or microcyst formation.
41% had syringomyelia associated with tonsillar
ectopia (Chiari I malformation), 28% had posttraumatic syrinx, 15% were associated with neoplasm,
and 15% were idiopathic. At least one third of patients with nonneoplastic cavities had associated
high-signal intensity in the cord contiguous to the cavity or in the parenchyma distal or proximal to
the cavity. This high-signal intensity in a simple
syrinx is speculated to represent gliosis, edema,
demyelination or microcyst formation.
![]() The
presence of edema is supported by the return of
the hyperintense parenchyma to normal signal
intensity following myelotomy and shunt
decompression of the cyst.
The
presence of edema is supported by the return of
the hyperintense parenchyma to normal signal
intensity following myelotomy and shunt
decompression of the cyst.
![]()
Examination of the entire spinal cord with sagittal images is mandatory. In regions where a cavity is suspected but somewhat equivocal in the sagittal projection, axial scans should be done. Failure to examine the entire spinal neuraxis may result in missing cavities that are separated from the primary or larger cavity. A simple (primary) syrinx must be distinguished from a cyst within a tumor.
Trauma
Of primary importance in spine trauma is the status of the cord. Cord injuries can have a variable
appearance. Severe injuries may reveal disruption of cord structure or even complete transection.
Milder contusions show high-signal edema within the cord on T2-weighted images, and perhaps cord
swelling.
![]() The presence of cord hemorrhage is associated with a poorer prognosis. In the setting of
new or progressive signs of neurologic dysfunction, MR can facilitate the diagnosis of delayed
complications, such as disk herniation, arachnoid cyst, and posttraumatic cord syrinx.
The presence of cord hemorrhage is associated with a poorer prognosis. In the setting of
new or progressive signs of neurologic dysfunction, MR can facilitate the diagnosis of delayed
complications, such as disk herniation, arachnoid cyst, and posttraumatic cord syrinx.
![]() The syrinx
or intramedullary cyst first forms at the site of injury but can extend some distance beyond and
become quite large. MR readily distinguishes between syrinx and other posttraumatic changes.
Myelomalacia and gliosis are focal processes with poorly defined margins and are associated with
cord atrophy. A syrinx has well-defined margins and frequently expands the cord.
The syrinx
or intramedullary cyst first forms at the site of injury but can extend some distance beyond and
become quite large. MR readily distinguishes between syrinx and other posttraumatic changes.
Myelomalacia and gliosis are focal processes with poorly defined margins and are associated with
cord atrophy. A syrinx has well-defined margins and frequently expands the cord.
Ischemia and Infarction
The spinal cord has a rather tenuous blood supply. A few radiculomedullary arteries contribute
to a single anterior and two small posterior spinal arteries. The anterior spinal artery supplies the
anterior two-thirds of the cord, including the anterior horns and corticospinal tracts. Causes of cord
ischemia and infarction include extrinsic compression from an epidural mass, surgical ligation of a
contributing intercostal artery, aortic aneurysm or dissection, emboli, hypotension, and spinal AV
malformation.
![]()
Spinal cord infarction causes focal edema and cord swelling. The T2 hyperintensity may exhibit an "owl's eye" appearance due to involvement of the central gray matter. With more severe ischemia, the entire cross section of the cord may be infarcted. Subacute infarcts may enhance with gadolinium. The chronic stage is featured primarily by cord atrophy.
Vascular Malformations
Spinal arteriovenous (AV) malformations can be dural or intradural. The dural variety is much more common (80% of cases) and most often occurs in the lower thoracic and lumbar regions. A low-flow fistula in the dura adjacent to a nerve root is supplied by a dural artery and drains into a medullary vein, causing venous congestion and ischemia of the spinal cord. Intradural AV malformations can be intramedullary or pial, are fed by the anterior spinal artery, and present with cord hemorrhage.
The intramedullary component of intradural AV malformations is generally readily visible on MR
images. Serpiginous flow voids and focal cord enlargement may highlight the site of the nidus.
Blood products from episodes of hemorrhage often give these lesions a heterogeneous texture. High
signal on T2-weighted images adjacent to the nidus may represent edema, ischemia, or gliosis.
![]() The
intramedullary component usually enhances with gadolinium.
The
intramedullary component usually enhances with gadolinium.
The dural AV malformations are more of a challenge. Diagnosis is often dependent on visual-izing the dilated veins on the surface of the cord. Sagittal T2-weighted images may show irregularity
of the cord surface, but flow artifacts can be problematic. A more reliable technique is a rapid set of
sagittal T1-weighted images following a bolus injection of gadolinium. The flow within the draining
veins is sufficiently slow to allow for intravascular enhancement.
![]()
Multiple Sclerosis
Spinal cord plaques are common and can be found at any level. Spinal cord lesions may be the
cause of the patient's presenting symptoms. Also, isolated cord involvement can occur. The MS
plaques are usually located in the dorsal and lateral aspects of the cord and often involve both gray
and white matter. They appear as elongated areas of high signal intensity on T2-weighted images and
are isointense to mildly hypointense on T1-weighted images. Acute plaques may produce cord
swelling and enhance with gadolinium.
![]()
Acute Transverse Myelopathy
Acute transverse myelopathy (myelitis) is an acute inflammation of one or more segments of the
spinal cord. It can be associated with multiple sclerosis, infections, vaccinations, systemic
malignancy, vascular disease, and autoimmune disorders. Acute lesions are hyperintense on T2-weighted images and hypointense on T1-weighted images and may demonstrate cord enlargement.
Enhancement with gadolinium has been reported. Chronic lesions often show persistent increased
T2 signal from residual gliosis. Severe cases may lead to cord atrophy.
![]()
INTRADURAL EXTRAMEDULLARY DISEASE
Tumors
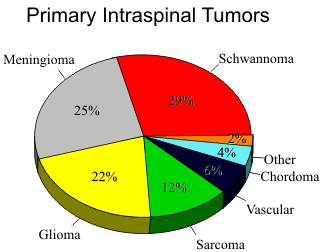
Analysis of large series of patients with intraspinal neoplasms has demonstrated a slightly higher incidence of nerve sheath tumors (35%) compared to meningiomas (25%). Distinction between the two groups of neoplasms is based the signal characteristics, location within the spinal canal, the presence of bone erosion, and growth patterns.
Nerve Sheath Tumors
Nerve sheath tumors include both neurofibroma and neurilemoma (schwannoma). They originate from Schwann cells of the myelin sheath that invest the nerve roots as they exit the spinal column. Schwannomas are usually solitary lesions and do not incorporate the nerve root. Neurofibromas are associated with neurofibromatosis and are generally multiple, and the nerve fibers become entangled within the tumor, making resection difficult without sacrificing the nerve. Nerve sheath tumors can be intradural, extradural or both, giving them a dumbbell appearance.
Bone erosion with scalloping of the
vertebral body margins or widening of the neural foramen with erosion of the pedicles is the hallmark
of the nerve sheath tumors. Even in the absence of bone erosion, extension of a soft-tissue mass
through the neural foramen with a dumbbell configuration is quite characteristic.
![]() The signal
characteristics of nerve sheath tumors are somewhat different from those of meningiomas. Nerve
sheath tumors typically have a T1 that is slightly longer than or equal to that of the spinal cord.
Lengthening of the TR and TE to increase T2-weighting results in brightening of the tumor. T2-weighted images, particularly with fat suppression, provide good contrast between the hyperintense
schwannoma, medium intensity paravertebral muscles, and hypointense fat.
The signal
characteristics of nerve sheath tumors are somewhat different from those of meningiomas. Nerve
sheath tumors typically have a T1 that is slightly longer than or equal to that of the spinal cord.
Lengthening of the TR and TE to increase T2-weighting results in brightening of the tumor. T2-weighted images, particularly with fat suppression, provide good contrast between the hyperintense
schwannoma, medium intensity paravertebral muscles, and hypointense fat.
Neurofibromas and schwannomas enhance brightly and uniformly with gadolinium. Enhancement is most helpful for intradural lesions, for detection of the smaller ones and definition of the larger ones. Gadolinium is less helpful for extradural lesions because the enhancing tumor becomes isointense to the surrounding fat. Fat suppression improves the contrast between enhancing extradural tumors and surrounding tissues.
Meningioma
Meningiomas arise from the meninges surrounding the spinal cord and are found primarily in the intradural extramedullary compartment. They are much more common in females and become symptomatic from cord compression during middle age or later. Over 80% occur in the thoracic region; another common site is the craniovertebral junction.
Meningiomas have many of the same imaging features as nerve sheath tumors. They often have similar signal characteristics on T1-weighted images, but as in the intracranial cavity, meningiomas tend to maintain signal intensity similar to that of the gray matter of the cord on T2-weighted images. Meningiomas also enhance rapidly and intensely, whereas neuromas take up the contrast more slowly. The presence of a broad base of attachment to a dural surface is a key feature of meningioma.
Metastatic Disease
Drop metastases generally originate from either primary, or less commonly, metastatic disease of the brain with subsequent seeding into the subarachnoid space. Because of either the dynamics of cerebrospinal fluid flow or gravitation, neoplastic cells implant on the spinal arachnoid or pia, resulting in a variety of appearances attributable to intradural metastases. Primary brain neoplasms that may lead to drop metastases include medulloblastoma, ependymoma, germinoma, choroid plexus carcinoma, teratoma, glioblastoma, and pineoblastoma. Secondary neoplasms metastatic to brain with a predilection for drop metastases include lymphoma, melanoma, and breast, lung and renal cell carcinoma. Intradural metastases may also occur from direct spread of extradural tumor or from lymphatic or hematogenous dissemination.
Several appearances of intradural tumor seeding have been noted. Most common are focal
nodular masses that may vary substantially in size, ranging from only a few millimeters to greater than
1 cm. They are frequently spread throughout the subarachnoid space, including laterally in the
recesses and root sleeves.
![]()
A second variety of intradural metastases is diffuse coating of the spinal cord with tumor, resulting in either smooth or nodular pseudo-expansion of the cord. This appearance may actually simulate an intramedullary lesion on routine non-contrast MR. The third expression of intradural metastatic disease is a homogeneous increase in signal within the subarachnoid space. This effect is seen predominantly in the lumbar region and is probably due to a combination of increased protein content within the CSF, malignant cells, and damping of CSF pulsations.
Gadolinium should be used in all cases of suspected intradural or "drop" metastases. It significantly increases the sensitivity for detection of the smaller nodules and readily distinguishes the pseudoexpansion described above from an intramedullary process.
Infection and Inflammation
Meningitis
Meningitis is an infection of the pia and arachnoid and adjacent cerebrospinal fluid. Bacterial infections generally run an acute course and do not elicit imaging changes in the spine. Fungal organisms and tuberculosis cause a more chronic inflammation and induce formation of a thick vascular granulation tissue. More unusal organisms can be encountered in the setting of immune suppression, particularly in patients with HIV infection.
Chronic spinal meningitis demonstrates enhancement of the leptomeninges. The enhancement is usually relatively smooth and of even thickness, lining the cord surface and dura. Chronic infections can lead to a myelitis, cord abscess, or a vasculits and cord infarction.
Sarcoidosis
Sarcoidosis is a granulomatous disease of unknown etiology. In approximately 5% of cases, the CNS is involved as a granulomatous infiltration of the leptomeninges. Intramedullary involvement can occur in severe cases. The meningeal inflammation enhances brightly with gadolinium. A nodular enhancement pattern usually distinguishes it from the infectious varieties.
CMV polyradiculopathy
In the setting of immune suppression, particularly in AIDS patients, CMV can produce a progressive polyradiculopathy. The clinical syndrome consists of progressive ascending weakness, lower extremity pain and sensory disturbance, saddle anesthesia, and urinary retention. Acute and chronic inflammation of the cauda equina and lumbosacral nerve roots results in destruction of axons and myelin. In severe cases the infection can ascend along the cauda equina to involve the conus medullaris.
Noncontrast MR scans are often negative. T1-weighted images with gadolinium reveal linear
enhancement of the nerve roots and the distal conus. Swelling and clumping of the nerve roots within
the thecal sac also occurs.
![]()
Guillain-Barré syndrome
The etiology for this acute polyneuropathy is unknown, but it is probably some type of autoimmune reaction to an antecedent viral infection. The Guillain-Barré syndrome consists of ascending motor weakness that can progress to total paralysis over a period of days to weeks. The illness has a monophasic course, and recovery is generally favorable in those patients that survive.
The MR manifestations are similar to CMV, namely diffuse enhancement of the cauda equina on
gadolinium-enhanced T1-weighted images. Involvement of the spinal cord may be more pronounced
in Guillain-Barré.
![]()
Arachnoiditis
One of the complications occurring after surgery, meningitis, subarachnoid hemorrhage, and
Pantopaque myelography is the development of arachnoiditis. Morphologic signs of arachnoiditis
include clumping of the nerve roots within the thecal sac with the creation of an intradural pseudomass. Adhesion of individual nerve roots to the inner margins of the arachnoid mater gives the
appearance of an empty thecal sac.
![]() The intradural cysts can be difficult to detect if their contents
have signal characteristics similar to those of CSF. If their protein content is increased, T1 shortening
results in increased signal on T1-weighted images. Damping of CSF pulsations may become apparent
on gradient-echo or T2-weighted sequences.
The intradural cysts can be difficult to detect if their contents
have signal characteristics similar to those of CSF. If their protein content is increased, T1 shortening
results in increased signal on T1-weighted images. Damping of CSF pulsations may become apparent
on gradient-echo or T2-weighted sequences.
REFERENCES